
Carbon is a non-metallic element with a chemical symbol of C. It is the fourth most abundant element in the universe, and the 15th most abundant element in the Earth’s crust. It is also the second most abundant element in humans after oxygen. Its chemical makeup leads to several unique properties of carbon.
Carbon belongs to Group 14 of the periodic table. Its atomic number is 6 and it has an atomic weight of 12.011. The oxidation states of carbon can vary from -4 to +4, where +4 exists in compounds such as methane and carbon disulfide, and +2 for carbon monoxide.
The physical properties of different carbon allotropes make them useful in batteries, electronics and nanomaterials. Carbon is also the “king of elements,” forming almost 10 million compounds to date including organic, inorganic and organometallic compounds.
Isotopes of carbon are extensively used for radiocarbon dating (carbon-14), molecular structure and medical research (carbon-13). In addition, carbon fibers show excellent mechanical properties and are popular in aerospace and civil engineering.
Allotropes of Carbon
Carbon has different allotropic forms, with diverse molecular configurations and atomic structures. The physical properties of carbon vary greatly with each allotrope. Some of the most well-known allotropes of carbon include graphite, diamond and fullerenes.
Graphite is one of the softest known materials and is used in pencils and as a solid lubricant. It is also a good conductor of electricity, making it useful in batteries and solar panels.
Graphene is simply one atomic layer of graphite arranged in a honeycomb lattice. In a graphene layer, each carbon atom is bonded covalently to three other atoms, leaving the fourth electron free to migrate in the plane, hence its electrical conductivity.
Diamond, on the contrary, is the hardest naturally occurring substance and is one of the unique properties of carbon. It has nearly twice the density of graphite, and each carbon atom is bonded tetrahedrally to four others with no free-flowing electrons. Thus diamond is a poor conductor of electricity. Diamond is also clear in appearance, unlike graphite, which is opaque.
Scientists have also synthesized other allotropes of carbon, such as fullerenes, carbon nanofoams and others. They possess special properties and constitute a blooming area of research in nanomaterials. Fullerenes are a group of hollow carbon molecules in a closed cage (buckyball) or cylinder (carbon nanotubes) conformation.
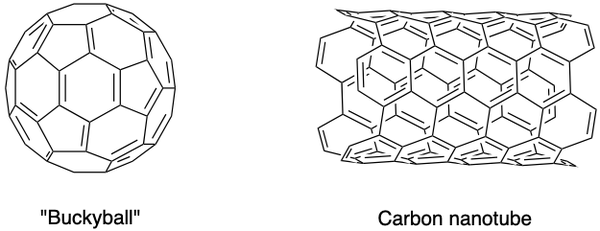
The C60 buckyball was discovered by Sir Harold Kroto, Richard Smalley and Robert Curl Jr., using a laser to vaporize graphite rods in a helium atmosphere. The carbon atoms are joined together by single and double bonds to form 12 pentagonal and 20 hexagonal faces in a soccer ball shape. Their pioneering efforts earned them the Nobel Prize in 1996.
Carbon nanotubes, which are elongated versions of buckyballs, were identified by Iijima Sumio. They are excellent conductors of heat and electricity and are useful for electronics.
Carbon nanotubes also demonstrate impressive tensile strength and have exciting applications in structural materials and medicine. However, the controlled synthesis of such nanomaterials has posed a great challenge to scientists.
Chemical Reactivity of Carbon
Carbon forms the basis of life on Earth, with millions of carbon-containing compounds making up 18 percent of all living things. It can form stable, covalent bonds with other atoms and appear as long chains or rings of strong interconnecting carbon-carbon bonds. These contribute to the diversity and complexity of carbon compounds existing on Earth.
These carbon compounds include organic molecules such as proteins, carbohydrates and DNA found in the cells of living organisms as well as inorganic compounds like carbon oxides. The study of organic molecules constitutes a specialized field called organic chemistry. Carbon can also form covalent bonds with metal as organometallic compounds. Iron porphyrin, which is the oxygen binding site of hemoglobin, is such an example.
Despite its abundance in nature, carbon is relatively unreactive under normal conditions. At standard temperature, it is not reactive toward acids (sulfuric acid or hydrochloric acid) or alkalis. It is also stable to oxidation at this temperature. At higher temperatures, however, carbon can react with oxygen to form carbon oxides (CO2 and CO), with sulfur gas to form carbon disulfide and with silicon to form carbides.
Isotopes of Carbon
There are 15 known isotopes of carbon, of which carbon-12 (98.93 percent of natural carbon) and carbon-13 (1.07 percent) are the two stable isotopes. Carbon-14 is the longest-lived isotope, with a half-life of 5,730 years. The shortest-lived carbon isotope is carbon-8, and it has a half-life of 1.98739 x 10−21 seconds.
The isotope carbon-14 is represented by 146C, where the presuperscript 14 is the atomic mass and the presubscript 6 is the atomic number. Carbon-14 has very low natural abundance (0.0000000001 percent), but its long half-life makes it useful for radiometric dating.
Carbon-14 is formed when nitrogen-14 reacts with neutrons from cosmic radiation, releasing a proton in this process. The carbon-14 then reacts with oxygen to generate 14CO2, which is evenly distributed in the atmosphere with 12CO2.
147N + 10n -------------> 146C + 11p
The carbon cycle starts when living organisms convert carbon dioxide (14CO2 and 12CO2 from the atmosphere) into organic compounds by photosynthesis and release it back to the atmosphere by respiration. In this equilibrium, there is a fixed ratio of 14CO2 and 12CO2 in the organisms. However when they die, the equilibrium stops, and the carbon-14 undergoes beta decay to the nitrogen-14 according to its 5,730 year half-life.
146C -------------> 147N + 0-1e
Measurement of the relative proportion of carbon-14 in a dead specimen thus allows calculation of the time elapsed after its death. This method of radiocarbon dating has been widely used for dating fossils and archaeological specimens from 500 to 50,000 years old.
Carbon-13 is another isotope that is used extensively in many applications. For instance, It is used in nuclear magnetic resonance (NMR) for determining molecular structures of organic compounds. It is also used as a labeling tool in combination with a mass spectrometer for medical research.
Mechanical Properties of Carbon Fibers
Carbon also exhibits useful mechanical properties, other than its physical, chemical and nuclear properties.
It can form alloys with steel into carbon steel, of which carbon content varies from 0.05 to 2 percent by weight. Medium-carbon steel (0.3-0.6 percent carbon) has balanced strength and ductility, as well as excellent tensile strength. Through a process of heat treating, ultra-high-carbon steel (1.25-2 percent carbon) can be tempered to great hardness and used for the manufacture of knifes.
Carbon fibers, which are 5 to 10 μm thick fibers composed mostly of carbon atoms, demonstrate high stiffness, tensile strength, chemical resistance, temperature tolerance and low weight and thermal expansion. The yield strength of steel is dependent on its grade and mild steel has a yield strength of 247 MPa. Carbon fibers have tensile strengths ranging from 1,600 to 6,370 MPa and are therefore popular in the field of aerospace, civil engineering and sports.
When a stress is exerted on a material, it deforms elastically at first. At this stage, it can return to its original shape when the stress is removed. Yield strength is defined as the stress a material can withstand without permanent deformation.
When it reaches a point (upper yield point) where it can no longer return to its original dimensions, it undergoes plastic deformation, which is a permanent and irreversible. Tensile strength is the maximum strength a material can withstand without failing or breaking.
References
About the Author
Lan Luo has a PhD in Organic Chemistry from University of Chicago and a BS in Chemistry from Worcester Polytechnic Institute. She has years of research experience in asymmetric catalysis, natural product synthesis, drug discovery and drug delivery. She has served as a contributor for Synfacts and a reviewer for journal articles.
