Batteries can be constructed using common household materials. It's all a matter of chemistry: When acids are present in a solution, ions are produced. When two dissimilar metals are introduced into the solution, an electric current forms between them, producing electricity. Create a bleach battery the next time you need a science fair project.
- Plastic cup
- Water
- Salt
- Vinegar
- Clorox bleach (or other household chlorine bleach)
- Pencil
- Two 12- to 18-inch pieces of 20-gauge insulated wire
- Wire strippers
- Aluminum foil
- Uncoated iron nail
- Multimeter
Try different metals in the bleach solution to see what difference you get in electricity output.
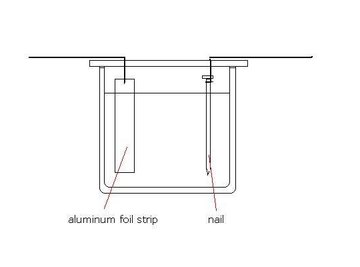
Fill a plastic or other nonmetallic cup about 2/3 to 3/4 full with tap water. Add the following: 1 teaspoon of salt, 1 teaspoon of vinegar and about 1 teaspoon of household bleach (Clorox Bleach works well).
Place a pencil or small dowel rod on the rim of the glass so that it makes a "bridge" that extends over the top of the cup.
Take two 12- to 18-inch pieces of insulated wire (20-gauge works well) and use wire strippers to remove about 1 inch of insulation from both ends of the wires.
Take one wire and wrap the bare end around the head end of a nail. Wrap the wire around the pencil so that the nail is suspended in the bleach solution.
Wrap the second wire onto a strip of aluminum foil. Then wrap the wire around the pencil so that the foil strip is suspended in the bleach solution.
Your battery is complete. To measure the voltage output, connect the wire ends to a multimeter. The output will be low, but in some cases, you may be able to power a small device with this battery or a series of interconnected batteries.
Things You'll Need
Tips
Tips
- Try different metals in the bleach solution to see what difference you get in electricity output.
About the Author
Based in Virginia, Nichole Liandi has been a freelance writer since 2005. Her articles have appeared on various print and online publications. Liandi has traveled extensively in Europe and East Asia and incorporates her experiences into her articles. She holds a Bachelor of Arts in history from West Virginia University.
Photo Credits
NLGraphics
