
The halogens are chemical elements that constitute Group 17 of the periodic table.
The six non-metallic elements are fluorine (F), chlorine (Cl), bromine (Br), iodine (I), astatine (As) and tennessine (Ts). The name “halogen” comes from a Greek word meaning “salt-producing,” since fluorine, chlorine and iodine produce a sea salt-like substance when they form a compound with an alkaline metal.
The characteristics of halogens are similar across the chemical series, although a progressive change in the properties is observed from fluorine through chlorine, bromine, iodine to astatine. In fact, fluorine is the most reactive element in the halogen family, exhibiting certain properties distinct from other halogens in the same family.
Chlorine gas (Cl2) is extensively used for water purification, and sodium chloride is the common table salt that you use every day. Fluorides are widely used in toothpastes to prevent tooth decay, while organic fluorides serve as refrigerants and lubricants. Bromine compounds are also used in pesticides, and iodine (I2) is a well-known antiseptic agent.
Atomic Characteristics of Halogens
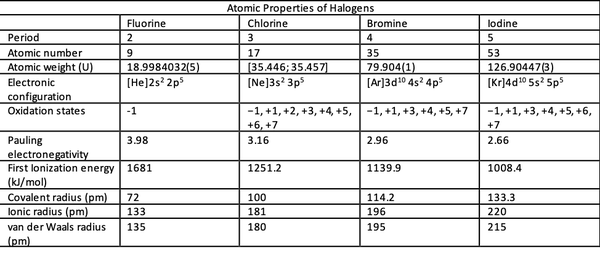
The halogen family occupies Group 17 (Group VII or VIIA) of the periodic table, and the group number designates the number of valence electrons. Halogens have seven valence electrons in their outermost shell, which can receive an electron from another atom to satisfy the octet rule and thus form stable compounds. Because of their great reactivity, halogens are found in nature not as the free elements, but rather as the diatomic molecules, for example Cl2 and I2.
Going down the rows in the periodic table, the energy level rises but not the electron configuration at the highest level. The valence electron configuration of fluorine is 2s2 2p5 while that of iodine is 5s2 5p5. The number of layers of electrons around the nucleus also increases from fluorine through chlorine, bromine to iodine. As a result, the atomic radius increase since more layers take up more space due to electron repulsion.
Electronegativity is a measure of the ability of an atom to attract electrons to itself. It is usually measured on the Pauling scale, and the values are determined experimentally. Fluorine has the highest electronegativity of 3.98 among halogens, and the electronegativity decreases down the group. Ionization energy is the amount of energy required to remove an electron from an atom. The more electronegative the element, the higher the ionization energy. Thus, fluorine has the highest ionization energy across the halogen family.
Physical Characteristics of Halogens
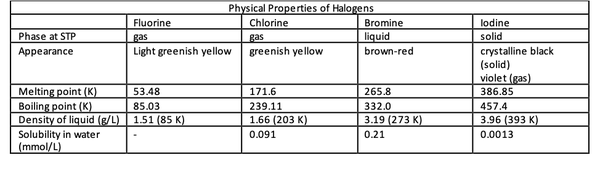
At standard conditions for temperature (273.15 K or 0 °C) and pressure (105 Pa or 1 bar), fluorine and chlorine are gases, bromine is a liquid, and iodine exists as a solid. As a general trend, the melting and boiling points increase down the halogen group. Since the halogens exist as diatomic molecules, van der Waals dispersion forces are the primary intermolecular forces among the molecules.
These forces are more prominent in larger molecules since there are more electrons moving around the nucleus to form temporary dipoles that create van der Waals forces.
Fluorine reacts violently with water to form hydrogen fluoride, oxygen and ozone. Chlorine dissolves slightly in water to produce a pale yellow solution. The aqueous solution of bromine adopts a range of colors from yellow to reddish-brown depending on the concentration. Iodine solution is pale brown, although its solid and gas are crystalline black and violet.
Chemical Properties of Halogens
Halogens are strong oxidizing agents. Since halogens have seven electrons in their valence shells, they have the tendency to take electrons from other molecules and thus oxidizing them. This oxidizing capability decreases down the halogen group, with fluorine the strongest oxidizing agent.
Oxidizing ability: F2 > Cl2 > Br2 > I2 > At2
For example, fluorine can oxidize water to oxygen and has the ability to replace any halide ions from its compounds, as shown in the following equations. Chlorine, however, only has the ability to take electrons from bromide, iodide and astatide ions, and bromine only iodide and astatide ions.
2F2 + H2O → 4HF + O2
F2 + 2Cl− → 2F− + Cl2
F2 + 2Br− → 2F− + Br2
F2 + 2I− → 2F− + I2
F2 + 2At− → 2F− + At2
For halide ions, the fluoride ion exhibits no reducing properties, while the iodide ion is a mild reducing agent. The trend in reducing ability increases from fluoride to iodide. This is demonstrated in the reaction of iodide with concentrated sulfuric acid, in which iodide reduces the sulfuric acid in three steps.
H2SO4 → SO2 → S → H2S
Sulfur oxidation state: +6 → +4 → 0 → -2
The oxidation state of sulfur is reduced from +6 in sulfuric acid to -2 in hydrogen sulfide. This reaction is exothermic, and gives off iodine (purple vapor) and hydrogen sulfide (“rotten egg” smell) as the major products.
H2SO4 + 8H+ + 8I− → 4I2 + H2S + 4H2O
The halogens also form compounds with hydrogen as hydrogen halides. The hydrogen-halogen bond decreases from fluoride to iodide, and hydrogen halide molecules form hydrogen bonds with each other in a zig-zag fashion. The acidity of the corresponding acid, when hydrogen halides are dissolved in water, increases from hydrofluoric acid to hydroiodic acid. Hydrobromic and hydroiodic acid are both strong acids. Although hydrofluoric acid is only a weak acid, it is similar in strength to organic acids like formic acid.
Uses of Halogens
Fluorine is used for the preparation of various fluorides. Hydrogen fluoride, when dissolved in water, produces hydrofluoric acid (HF). Despite being a weak acid, the fluoride anions in hydrofluoric acid can form a strong bond with the silicon molecules in glass.
Hydrofluoric acid is therefore widely used in industry for frosting and etching glass. It also acts as a metal pickling agent and etchant by removing oxides and other impurities from stainless steels. However, hydrofluoric acid is extremely corrosive, and it can cause poisoning and bone damage through the exposure of skin or eyes.
In the early 20th century, high concentration of fluoride in drinking water was associated with a much lower incidence of tooth decay. Although the use of fluoride in drinking water to improve overall dental health was controversial, fluoride had meanwhile gained popularity in toothpastes.
Colgate introduced “stannous fluoride” in 1955. Today, there are three types of fluorides approved by the FDA to prevent tooth decay: stannous fluoride, sodium fluoride and sodium monofluorophosphate.
Elemental fluorine can also react with hydrocarbons to form fluorocarbons, in which the hydrogen has been partially or fully replaced by fluorine, resulting in compounds that have excellent physical and chemical properties such as chemical inertness and high electrical resistance.
Polytetrafluoroethylene ((CF2CF2)x, known as PTFE or Teflon) is made from unsaturated fluorocarbons, and it provides a non-stick surface for cookware. Chlorofluorocarbons (CFCs) such as dichlorodifluoromethane (Cl2CF2) have been widely used as household refrigerants, but it plays an active role in ozone depletion. Its production was banned in 1996 by the Montreal Protocol, and hydrochlorofluorocarbons (HCFCs) have been used as substitutes.
Fluorine is also used in drug design and development as a means of enhancing biological activity and increasing chemical or metabolic stability. Fluoxetine, which is an antidepressant of the selective serotonin reuptake inhibitor (SSRI) class, is such an example with a trifluoromethyl group in the molecule.
Chlorine gas (Cl2) was used in World War I as a chemical weapon, since it is highly irritant to the respiratory system. Its solution was used as a disinfectant to combat a cholera epidemic that swept across Europe in 1831.
Sodium chloride (NaCl), or table salt, is one the most ubiquitous seasonings. It not only satisfies our culinary needs, but also provides essential nutrients for metabolism. Chlorine participates in the production of hydrochloric acid in the stomach and also serves important roles in cellular pump functions. Too little chloride in the diet can result in electrolyte disturbances like hypochloremia.
Another well-known use of chlorine is as bleach for disinfection and sanitization purposes. The active ingredient of bleaching powder (Ca(OCl)2∙CaCl2∙Ca(OH)2∙2H2O) is calcium hypochlorite (Ca(OCl)2), and it has been used for disinfecting swimming pools.
Chlorine can react with carbon monoxide to form carbonyl chloride (COCl2) or phosgene, which is used for synthesizing isocyanates and polyurethanes and in metallurgy to convert oxides into chlorides. Chlorine also reacts with sulfur dioxide to form thionyl chloride (SO2Cl2), which is a common chlorinating reagent in organic synthesis and is used in the vulcanization of rubber.
CO + Cl2 → COCl2
SO2 + Cl2 → SO2Cl2
Chlorine is also frequently seen in organic compounds, such as polyvinyl chloride (PVC) and dichlorodiphenyltrichloroethane (DDT). It has great importance in the chemical, material and pharmaceutical industry.
The earliest use of bromine is seen in tyrian purple, which is a purple dye made from murex mollusks by the Phoenicians. Bromine is still used in dyes such as brominated anthraquinone derivatives.
At one time, ethylene dibromide (C2H4Br2) was widely used in in the petroleum industry. It was added to gasoline to prevent deposition of lead in the engine. However, leaded gasoline was phased out in the 1970s. Ethylene dibromide is still used today as a pesticide to destroy nematodes and pests in soils.
In the early 20th century, bromides of potassium, sodium and calcium were used as sedatives in medicine. Brominated flame retardants (BRFs) can reduce the flammability of the products containing them, such as protectors or covers for electrical appliances. Similar to silver chloride and iodide, which are light sensitive, silver bromide (AgBr) is an important component of photographic film.
The best-known use of iodine is its importance in human diet. In the thyroid gland, iodine is converted to thyroxine, which is a hormone necessary for metabolism. An iodine deficiency will often lead to a goiter (enlargement of the thyroid) or myxedema. Potassium iodide (KI) is frequently added to table salt to protect against this deficiency. Iodine can also be supplemented by consuming iodine-rich food products such as seaweed.
Iodine is widely used as a disinfectant and antiseptic, often as a solution of potassium iodide. Iodine is also used in early photography: The daguerreotype process uses silver plates sensitized with gaseous iodine. Iodine compounds are used extensively in chemical analysis.
Iodometry measures the release or uptake of iodine base on the titration of iodine with sodium thiosulfate. Iodine compounds are also employed as catalysts for certain organic reactions.
References
About the Author
Lan Luo has a PhD in Organic Chemistry from University of Chicago and a BS in Chemistry from Worcester Polytechnic Institute. She has years of research experience in asymmetric catalysis, natural product synthesis, drug discovery and drug delivery. She has served as a contributor for Synfacts and a reviewer for journal articles.
