Lipids are organic compounds (chemicals built of the element carbon). In addition to carbon atoms, they consist mostly hydrogen, with a relatively low content of oxygen, nitrogen and other elements, as compared to other compounds found in biology. They are hydrophobic, meaning insoluble in water. In some cases, however, they can include hydrophillic--"water-loving" entities--such as phosphate groups, in which case they can dissolve both in oil and water. Types of lipids include oils and fats, waxes, steroids, phospholipids and similar compounds.
Oils and Fats
Oils and fats have the same chemical arrangement but differ functionally in that oils are liquid at room temperature, while fats are solid. The structure consists of a glycerol "backbone" to which three fatty acids are attached. A fatty acid is a chain of carbon and hydrogen atoms with a "carboxyl head," an entity made of a carbon and two oxygen atoms, giving it special properties. It is through the carboxyl head that the fatty acids attach to the glycerol via ester linkage. With all three carbons of the glycerol linked to a fatty acid, the compound is known as a "triglyceride."
Saturated, Mono-Unsaturated and Poly-Unsaturated
The term "saturation" refers to the number of "double bonds" in the hydrocarbon chain of a fatty acid, and this relates to the number of hydrogen atoms. As opposed to the more common single bond, a double bond between two carbon atoms is more rigid. By inhibiting rotation between the atoms, and drawing them close together, double bonds place kinks in a fatty acid chain. Triglycerides with fatty acids containing more double bonds tend to be liquid at room temperature, and thus are oils. If there is one double bond, a fatty acid is called "mono-unsaturated"; if there are many, it is called "poly-unsaturated."
Sources of Oils and Fats
Most oils come from plants and are largely unsaturated, although certain plant oils, such as palm and coconut, are highly saturated. Fats, on the other hand, tend to come from animals and have more saturated fatty acids.
Trans Fats
Unsaturated fatty acids can be saturated, and more often "partially saturated," through industrial processing. When fatty acids are partially saturated, the double bonds fare different from the kind that usually occur in nature. They are called "trans" double bonds, and fats that contain them are called "trans fats." Not only does the presence of trans fats in foods have implications for the melting point--and thus for cardiovascular health--but evidence suggests dietary trans fats may contribute to cancer.
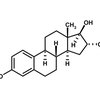
Sources of Waxes, Cholesterol and Steroids
Wax sources include bees, plants such as jojoba (Simmondsia chinensis) and carnauba palm (Copernicia cerifera), and sheep, whose wool contains the wax lanolin. Cholesterol comes from animal tissue, found in cell membranes and as a parent compound for a variety of steroid hormones. While waxes are long hydrocarbon chains with one or two functional groups, cholesterol and its steroid derivatives contain various hydrocarbon ring structures.
Phospholipids
Forming the bulk of cell membranes, phospolipids are similar to tryglycerides, except with one of the three fatty acids replaced with a phosphate group to which other highly polar (aka hydrophillic, water-loving) entities are attached. The two hydrocarbon chains, being hydrophobic, mix well with fat, while the polar heads of the compounds mix with water. This allows phospholipids to serve as carriers in blood for hydrophobic compounds like cholesterol, or to line up into membranes around cells.
References
Writer Bio
David Warmflash is an astrobiologist-writer, with a passion for communicating science to the general public. He serves as lead investigator for the Living Interplanetary Flight Experiment (LIFE), a Planetary Society-sponsored project, scheduled for launch in 2011 on the Russian Space Agency's Phobos-Grunt probe. Additionally, he is fascinated with ancient history.
Image Credit
Wavebreakmedia Ltd/Wavebreak Media/Getty Images